 In 1960 a revised metric system based on seven fundamental units was established, and the resulting SI unit of energy is the joule (J). Note that this calorie is not the same as the Calorie used for nutritional purposes. The traditional unit of heat energy used by organic chemists is the calorie, defined as the heat needed to raise the temperature of 1 gram of water 1º C (from 14.5 to 15.5º C). Of course, reactions that consume a gas undergo an opposite volume change, and work is done on the system by the surroundings. Also, if gases are produced in a reaction, the work done on the surroundings by volume expansion becomes part of the energy balance, and in explosive reactions may be the major consequence. Heat is the most common form of energy introduced into or released from chemical reactions, but light, electrical current and sound may also be involved. Chemical reactions involve the making and breaking of covalent bonds, and these transformations have energy consequences. In these examples the potential and kinetic energy are directed (oriented) in space, whereas the heat produced consists of random molecular motion.Ĭhemists are concerned with both physical and chemical change, and the energy associated with each. The accumulated snow on a steep mountain has considerable potential energy which is transformed in an avalanche, first into kinetic energy, and then into heat and work as the avalanche engulfs forests and towns and eventually comes to a stop. A speeding automobile has considerable kinetic energy, which is transformed into heat energy and work (the rupture of metal and plastic components) if the automobile crashes into a stone wall. Some of these chemicals gradually decompose giving off gas, while some may crystallize below certain temperatures, concentrating them.The term energy is commonly used in many contexts, but seldom in a carefully defined way. Azo compounds may have an SADT or a temperature below which storage can be hazardous. Some self-reactive chemicals can be dangerously reactive and can decompose when exposed to sunlight, heat, friction, or shock. The explosion hazard associated with some of these chemicals ranges from thermal explosion to deflagration or detonation. The primary hazards associated with self-reactive chemicals are fire and explosion. In laboratory testing, it neither detonates in the cavitated state nor deflagrates at all and shows no effect when heated under confinement nor any explosive power, provided it is thermally stable. In laboratory testing, it neither detonates in the cavitated state nor deflagrates at all and shows only a low or no effect when heated under confinement, as well as low or no explosive power. In laboratory testing, it neither detonates nor deflagrates at all and shows low or no effect when heated under confinement. Does not detonate or deflagrate at all, and shows a medium effect when heated under confinement.Does not detonate at all, deflagrates slowly, and shows no violent effect when heated under confinement. 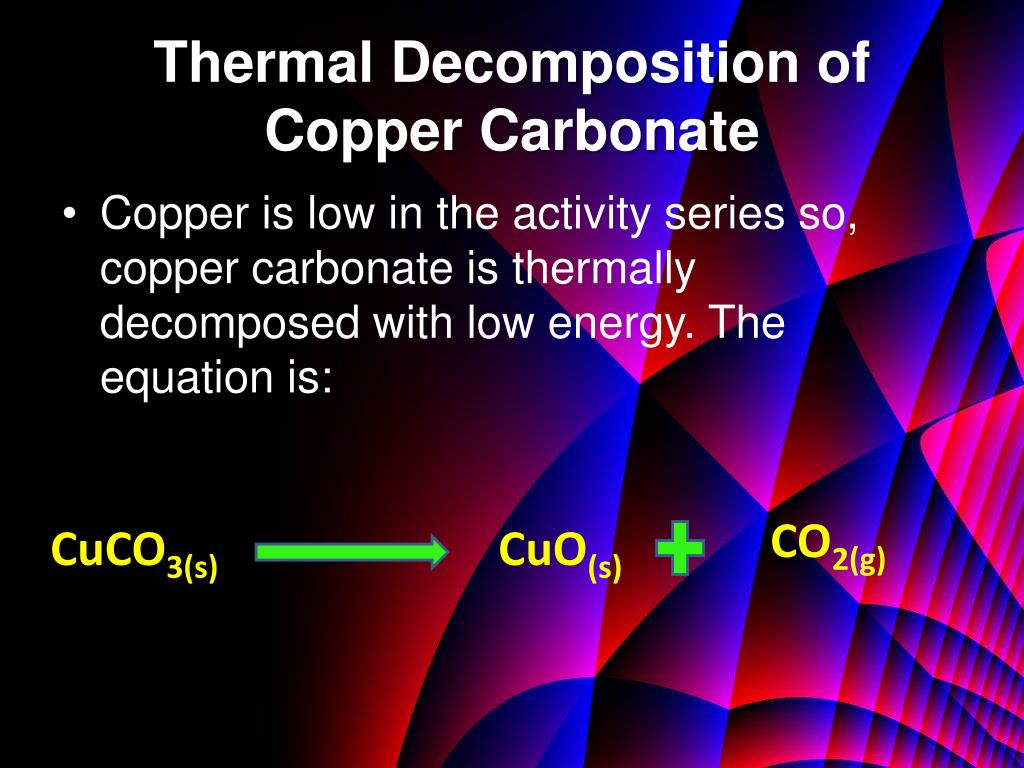
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/simple-experiment-58b5b3325f9b586046bbfa7f.jpg)
 RSS Feed
RSS Feed
